|
8/26/2023 0 Comments Si and potassium element 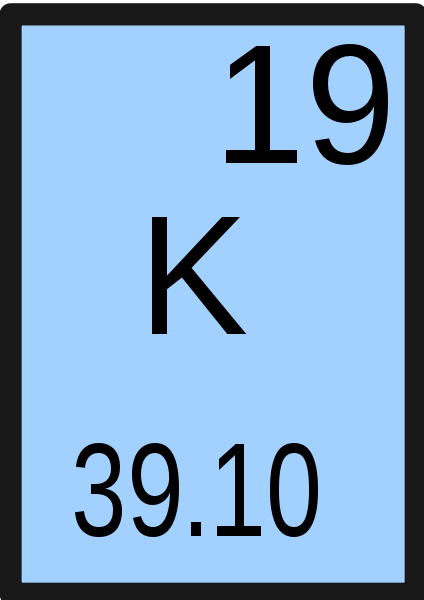
Metallic potassium is available commercially for about $40/lb in small quantities. The radioactivity presents no appreciable hazard. The influence of mineral substrate transformation is analyzed on the content of chemical elements in plants tomato, and wheat. Also, shells dont stack neatly one on top of another, so dont always assume an elements valence is determined by the number of electrons in its outer shell. Ordinary potassium is composed of three isotopes, one of which is 40 oK (0.0118%), a radioactive isotope with a half-life of 1.28 x 10 9 years. Remember that an elements electron cloud will become more stable by filling, emptying, or half-filling the shell. Seventeen isotopes of potassium are known. Potassium and its salts impart a violet color to flames. It rapidly oxidizes in air and must be preserved in a mineral oil such as kerosene.Īs with other metals of the alkali group, it decomposes in water with the evolution of hydrogen. It is soft, easily cut with a knife, and is silvery in appearance immediately after a fresh surface is exposed. Except for lithium, it is the lightest known metal. It is one of the most reactive and electropositive of metals. Many potassium salts are of utmost importance, including the hydroxide, nitrate, carbonate, chloride, chlorate, bromide, iodide, cyanide, sulfate, chromate, and dichromate. Potassium is an essential constituent for plant growth and is found in most soils.Īn alloy of sodium and potassium (NaK) is used as a heat-transfer medium. The greatest demand for potash has been in its use for fertilizers. Thermal methods also are commonly used to produce potassium (such as by reduction of potassium compounds with CaC 2, C, Si, or Na). Potassium is never found free in nature, but is obtained by electrolysis of the hydroxide, much in the same manner as prepared by Davy. The temperature dependence of the solubility of both elements was followed. Potassium is also found in the ocean, but is present only in relatively small amounts, compared to sodium. sodium or potassium into silicon crystals during the electrolysis of molten. Au, One distinguishing physical property of the element mercury is. True False, Select the correct symbol for the element named: sodium a. Large deposits of potash, found at a depth of some 3000 ft in Saskatchewan, promise to be important in coming years. Study with Quizlet and memorize flashcards containing terms like Mercury is a metal. Potash is mined in Germany, New Mexico, California, Utah, and elsewhere. Most potassium minerals are insoluble and the metal is obtained from them only with great difficulty.Ĭertain minerals, however, such as sylvite, carnallite, langbeinite, and polyhalite are found in ancientlake and sea beds and form rather extensive deposits from which potassium and its salts can readily be obtained. The metal is the seventh most abundant and makes up about 2.4% by weight of the earth's crust. kalium, Arab qali, alkali) Discovered in 1807 by Davy, who obtained it from caustic potash (KOH) this was the first metal isolated by electrolysis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
